Tantalum boride
In today's article, we are going to explore Tantalum boride and its relevance in modern society. From its impact on people's daily lives to its influence in the professional field, Tantalum boride has become a topic of great interest in recent years. As we progress through this article, we are going to take a closer look at how Tantalum boride has evolved over time and the implications it has on different aspects of our lives. Additionally, we will also discuss the future prospects of Tantalum boride and how it can continue to shape our world in the years to come. Get ready to immerse yourself in the fascinating world of Tantalum boride!

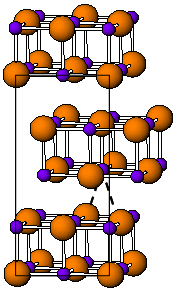
Tantalum borides are compounds of tantalum and boron most remarkable for their extreme hardness.
Properties
The Vickers hardness of TaB and TaB2 films and crystals is ~30 GPa.[1][2][3] Those materials are stable to oxidation below 700 °C and to acid corrosion.[1][3]
TaB2 has the same hexagonal structure as most diborides (AlB2, MgB2, etc.).[4] The mentioned borides have the following space groups: TaB (orthorhombic, Thallium(I) iodide-type, Cmcm), Ta5B6 (Cmmm), Ta3B4 (Immm), TaB2 (hexagonal, aluminum diboride-type, P6/mmm).[3]
Preparation
Single crystals of TaB, Ta5B6, Ta3B4 or TaB2 (about 1 cm diameter, 6 cm length) can be produced by the floating zone method.[2][3]
Tantalum boride films can be deposited from a gas mixture of TaCl5-BCl3-H2-Ar in the temperature range 540–800 °C. TaB2 (single-phase) is deposited at a source gas flow ratio (BCl3/TaCl5) of six and a temperature above 600 °C. TaB (single-phase) is deposited at BCl3/TaCl5 = 2–4 and T = 600–700 °C.[1]
Nanocrystals of TaB2 were successfully synthesized by the reduction of Ta2O5 with NaBH4 using a molar ratio M:B of 1:4 at 700-900 °C for 30 min under argon flow.[5]
- Ta2O5 + 6.5 NaBH4 → 2 TaB2 + 4 Na(g,l) + 2.5 NaBO2+ 13 H2(g)
References
- ^ a b c Motojima, Seiji; Kito, Kazuhito; Sugiyama, Kohzo (1982). "Low-temperature deposition of TaB and TaB2 by chemical vapor deposition". Journal of Nuclear Materials. 105 (2–3). Elsevier BV: 262–268. Bibcode:1982JNuM..105..262M. doi:10.1016/0022-3115(82)90383-x. ISSN 0022-3115.
- ^ a b Otani, S; Korsukova, M.M; Mitsuhashi, T (1998). "Floating zone growth and high-temperature hardness of NbB2 and TaB2 single crystals". Journal of Crystal Growth. 194 (3–4). Elsevier BV: 430–433. Bibcode:1998JCrGr.194..430O. doi:10.1016/s0022-0248(98)00691-5. ISSN 0022-0248.
- ^ a b c d Okada, Shigeru; Kudou, Kunio; Higashi, Iwarni; Lundström, Torsten (1993). "Single crystals of TaB, Ta5B6, Ta3B4 and TAB2, as obtained from high-temperature metal solutions, and their properties". Journal of Crystal Growth. 128 (1–4). Elsevier BV: 1120–1124. Bibcode:1993JCrGr.128.1120O. doi:10.1016/s0022-0248(07)80109-6. ISSN 0022-0248.
- ^ Chen, Xing-Qiu; Fu, C. L.; Krčmar, M.; Painter, G. S. (2008-05-16). "Electronic and Structural Origin of Ultraincompressibility of5dTransition-Metal DiboridesMB2(M=W, Re, Os)". Physical Review Letters. 100 (19). American Physical Society (APS): 196403. Bibcode:2008PhRvL.100s6403C. doi:10.1103/physrevlett.100.196403. ISSN 0031-9007. PMID 18518467.
- ^ Zoli, Luca; Galizia, Pietro; Silvestroni, Laura; Sciti, Diletta (23 January 2018). "Synthesis of group IV and V metal diboride nanocrystals via borothermal reduction with sodium borohydride". Journal of the American Ceramic Society. 101 (6): 2627–2637. doi:10.1111/jace.15401.